Nanopore Sequencing (ONT)
Nanopore sequencing developed by Oxford Nanopore Technologies (ONT) uses molecular sensors or nanopores capable of measuring changes in electric current when a single DNA or RNA molecule passes through them. Different sequences going through a nanopore give different electrical signals, producing an electrical trace or ‘squiggle.’ These squiggles can be decoded into basecalls in real-time. Read lengths produced by Nanopore sequencing are only limited by the size of the library, which enables obtaining DNA reads spanning up to 2Mb and full-length transcripts. Nanopore technology can sequence native DNA and RNA molecules, allowing for the detection of base modifications. The PromethION sequencer is the highest throughput Sequencer from ONT. Each PromethION flow cell is advertised to produce up to 180Gb1 of data.
ONT - Client-made library sequencing service »
General guidelines for sequencing your ONT library with us
ONT - Genomic DNA service »
General guidelines on the Genomic DNA sequencing service using ONT technology at the BRF
Sequencing applications supported by the ONT sequencing include:
- Structural variation
- Assembly
- DNA and RNA base modifications
- Full length transcriptome
- Multiplex sequencing
1. Check that your samples meet submission criteria
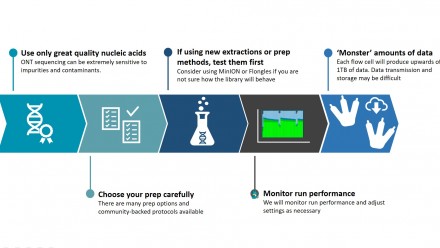
- Refer to the appropriate guide for the amount, concentration, size profile and volume required for the service. For example, for gDNA submission, use the gDNA_ONT guide
2. Fill in the sample submission form in IdeaElan
- Log into IdeaElan and ensure you have access to the BRF Shared Equipment and Store Facility
- Locate the ONT_gDNA form under Request Service > Sample Submission form
- Complete the form with your project details. If you are unsure, plese feel free to get in touch via email or phone.
3. Prepare your samples and bring them to the BRF
- Include a printed copy of the electronic submission form with your samples.
- Submit your samples using 1.5mL microfuge tubes, preferably eppendorf lo-bind.
- Label your tubes with the Sample submission ID (e.g ONT_gDNA) and the Request Id (consecutive numbers from 1) from the "Samples" section of the sample submission form.
- Notify us by email before you intend to bring your samples. If you are shipping, please provide us with an approximate delivery date.
4. Verify if you want to proceed with processing or withdraw your submission.
- Upon reception of your samples, we will conduct quality checks and send you the results to review. Based on these, you will be asked if you wish to proceed with or withdraw your project.
Terms and Conditions of Service - ONT Sequencing
The use of the BRF Oxford Nanopore Technologies (ONT) genomic services at the BRF are ruled by the following conditions:
Services:
- The BRF agrees to perform the services for you (the Client) with due care and skill, and to include all applicable quality control checks throughout sample processing to ensure manufacturer’s specifications are met. The Client acknowledges and agrees that the BRF is not responsible for the data output/quality from:
- Samples that do not meet BRF quality control checks as per sample submission guides.
- Libraries prepared by the Client.
- Libraries derived from assays that deviate from recommended manufacturer’s protocols, if those deviations are approved by the Client.
- Turnaround times indicated verbally or in writing by BRF staff are on a ‘reasonable endeavours’ basis and not a binding commitment.
- The BRF reserves the right to refuse any requests for Service at our discretion.
- The BRF owns all intermediate and end products derived during the processing of Samples including libraries.
- The Client acknowledges that ONT data generated by the BRF is for research purposes only and will not be used:
- As part of any non-research related purpose, including commercial, clinical, diagnostic or therapeutic procedures.
- For the development of any other product or service that competes or could compete with the ONT products or services
- For monitoring, benchmarking or designing products to better compete with the ONT products or services.
Samples:
- The Client will only submit highly purified nucleic acids that are non-hazardous and non-infectious for ONT sequencing.
- Submitted samples should be de-identified and do not contain any personal information.
- The Client agrees to follow sample submission guidelines, including completing the BRF’s electronic sample submission form and meeting sample acceptance criteria as per service guidelines (details of the quality and quantity required are on the BRF website and sample submission form). If the project does not comply with acceptance criteria, the Client will:
- Submit replacement samples or
- withdraw the submission
- Pay the BRF for failed quality checks performed on the samples.
- Agree to cover the cost of any consumables ordered on their behalf if the supplied sample does not meet sample acceptance criteria.
- The Client may arrange for failed/left over samples to be returned, at the Client expense. The Client acknowledges that the BRF may discard rejected/leftover samples after 30 days of the project closure/completion.
Consumables and flow cells:
- Upon receipt of consumables, the Client accepts responsibility for the correct handling, use, storage and disposal of the consumables and flow cells.
- The Client agrees to return any used flow cell purchased through the BRF.
- The BRF extends no warranties of any kind in respect to the consumables. Any consumables sold may have hazardous properties. The Client agrees to use appropriate caution and safeguards as not all properties are known.
- The Client accepts to use consumables and flow cells in accordance with the Nanopore Device Purchase Terms and Conditions (ONT Agreement). ONT specifically excludes the use of consumables and flow cells for the following purposes:
- Use of consumables and/or flow cells for clinical, diagnostic or therapeutic procedures where validation or registration with regulatory authorities is required.
- Use of the consumables and/or flow cells for commercial or other non-research purposes.
- To generate ONT sequencing libraries in conjunction with reagents or consumables not sold or approved by ONT
- Use ONT Consumables in conjunction with non-ONT hardware, flow cells or devices;
- Modify, copy or create any derivative works of the consumables (except to the extent applicable laws specifically prohibit such restriction)
- Reverse engineer, decompile, disassemble or otherwise attempt to derive the composition of the consumables and flow cells
- Redistribute, encumber, sell, rent, lease, sublicense or otherwise transfer rights to the consumables and flow cells
- Other exclusions as per ONT Agreement.
- The BRF will not be liable to the Client for any loss, claim or demand made by the Client due to acceptance, handling, use, storage or disposal of consumables and/or flow cells by the Client, except to the extent permitted by law when it is the result of wilful misconduct on the part of the BRF or its employees.
Data retention:
- Clients are responsible for archiving and safely storing data generated by the BRF. ONT Data will, upon confirmation of reception, immediately become the responsibility of the Client. The Client agrees to:
- Notify the BRF that ONT data has been received without errors within 1 week of data handover.
- When data reception notification is not provided to the BRF by the Client within 1 week, the Client agrees to pay a storage fee for each week data reception notification is not given to the BRF. The BRF will archive data for a maximum of 3 months. If the Client requires Data to be recovered from the BRF archive, a recovery fee will apply.
- The BRF reserves the right to permanently delete data upon receiving confirmation of data reception by the Client or after 3 months of data handover if confirmation is not received.
- The BRF will not be liable to the Client for any loss, claim or demand made by the Client due to acceptance, handling, use, storage or deletion or data by the Client.
Fees and other expenses:
- BRF’s ONT service prices are subjected to change at short notice. Prices offered to Clients will only be maintained for 30 days after enquiry for the service is made or a quote is generated. The Client has to provide samples of adequate quality and quantity within those 30 days to maintain the offered price.
- The BRF reserves the right to review processing costs if:
- The number of samples changes during the life of the project
- The assays/procedures agreed upon or quoted change from the time of project acceptance
- Sample submissions need to be modified to comply with project acceptance requirements
- Data reception is not acknowledged by Client within one week of handover, which may incur in data archiving fees.
Acknowledging the BRF:
- The BRF operates on a cost recovery basis. The percentage of subsidisation is determined by JCSMR.
- No samples or orders will be processed without an authorised* sample submission/order form and valid charge code. (*Authorised by the signature of the PI/Lab Head, or by granting of electronic access to BRF ordering systems with the authority of the PI/Lab Head).
- All work performed in the BRF, as well as the use of any BRF resources should be acknowledged in all publications and presentations arising from that work, both in the Methods and in the Acknowledgement sections. Mentions and acknowledgments shall also state that NCRIS-enabled Bioplatforms Australia infrastructure were used for the work. Examples include:
- In the Methods Section: “We sequenced our sample on a ONT PromethION Sequencer using NCRIS-enabled Bioplatforms Australia infrastructure at the ACRF Biomolecular resource Facility at the John Curtin School of Medical research”
- In the Acknowledgements section: “This study/research used NCRIS-enabled Bioplatforms Australia infrastructure and we thank the Biomolecular Resource Facility at the John Curtin School of Medical Research for performing the method...”
- The client agrees to share a reference any publication, including theses, with the Manager of the BRF once the paper is published. Contact details can be found in the BRF’s website.
- Although the BRF is primarily a service unit, there are instances where BRF staff make significant contributions to either the technical or intellectual input of the project. Acknowledgement of individual BRF staff as collaborators or co-authors are solely at the Client’s discretion, but we encourage the client to discuss this with the BRF at the start of the project.