Chromatin and RNA surveillance
Although only a small percentage of our genome consists of protein coding genes, the majority of the genome is transcriptionally active, resulting in significant quantities of non-coding RNA (ncRNA) transcripts. Most ncRNAs, so-called cryptic unstable transcripts (CUTs), are rapidly degraded by the nuclear exosome. However, the way these CUTs are recognised and targeted to the exosome is not well understood. CUTs are generally very similar to mRNA transcripts (e.g., capped and poly-adenylated), therefore recognition and sorting of different classes of RNAs must occur co-transcriptionally by recruiting various RNA binding complexes to the nascent transcripts. While mRNAs are packaged and exported to the cytoplasm, CUTs are loaded with RNA surveillance factors that deliver these RNAs to the degradation machinery. The mechanism by which the RNA polymerase II (RNAPII) machinery distinguishes between the different types of RNA transcripts is not well understood, but emerging evidence shows that epigenetic modifications and the chromatin environment of the transcribed DNA region play a major role in this process. We are studying the co-transcriptional recruitment of RNA binding complexes to the nascent transcripts, especially focusing on the crosstalk between chromatin modifications and RNA processing.
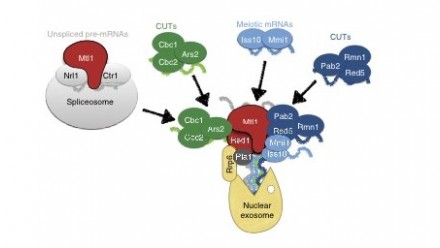
We recently identified the multi-subunit MTREC complex in S. pombe that specifically binds to CUTs and unspliced pre-mRNAs and targets them to the nuclear exosome for degradation (Zhou et al., 2015). Phylogenetic analysis of the subunits shows high similarity to subunits of the human Nuclear EXosome Targeting (NEXT) complex. Deletion or mutation of S. pombe MTREC complex subunits leads to strong accumulation of CUTs and unspliced or mis-spliced pre-mRNAs. MTREC complex physically interacts with the nuclear exosome, and with various RNA binding and processing complexes, including the cap-binding, spliceosome, cleavage and poly-adenylation complexes. Our future goal is to further characterize the MTREC complex in vitro and in vivo, and to uncover the molecular mechanism by which this large, multi- subdomain complex is recruited to CUTs and aberrant mRNAs and targets them to the exosome.
IMAGE CAPTION: Proposed model for the role of MTREC complex in RNA surveillance. Submodules of the MTREC complex, together with the Mtl1–Ctr1–Nrl1 complex, are recruited to different subsets of CUTs, meiotic mRNAs or unspliced pre-mRNA transcripts and deliver these RNAs to the MTREC complex. The RNAs are polyadenylated by the canonical poly(A) polymerase, Pla1. The RNA-loaded MTREC complex can dock to the nuclear exosome through the Red1–Rrp6 interaction. The helicase activity of the Mtl1 subunit then feeds the MTREC-bound RNAs into the exosome channel.